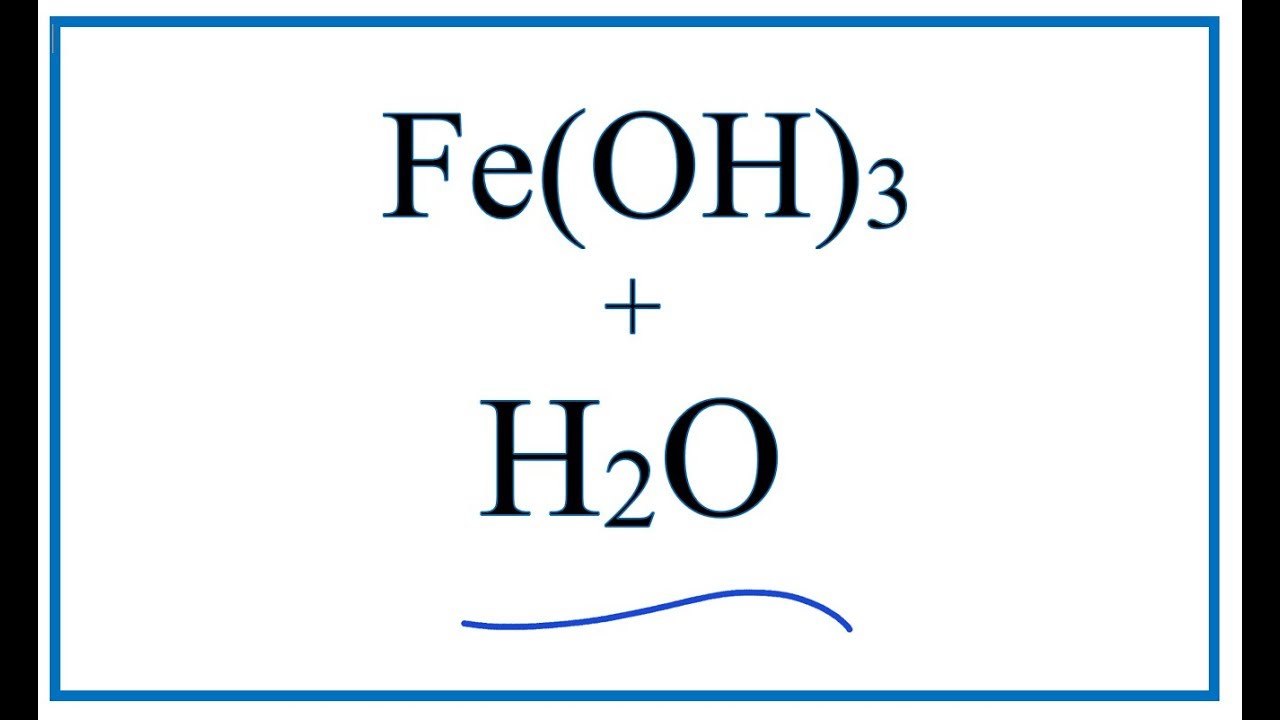
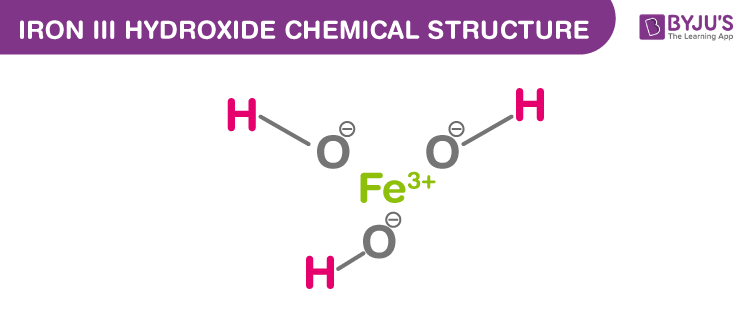
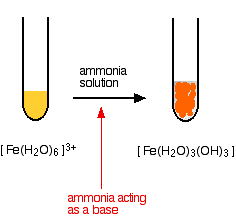
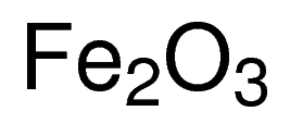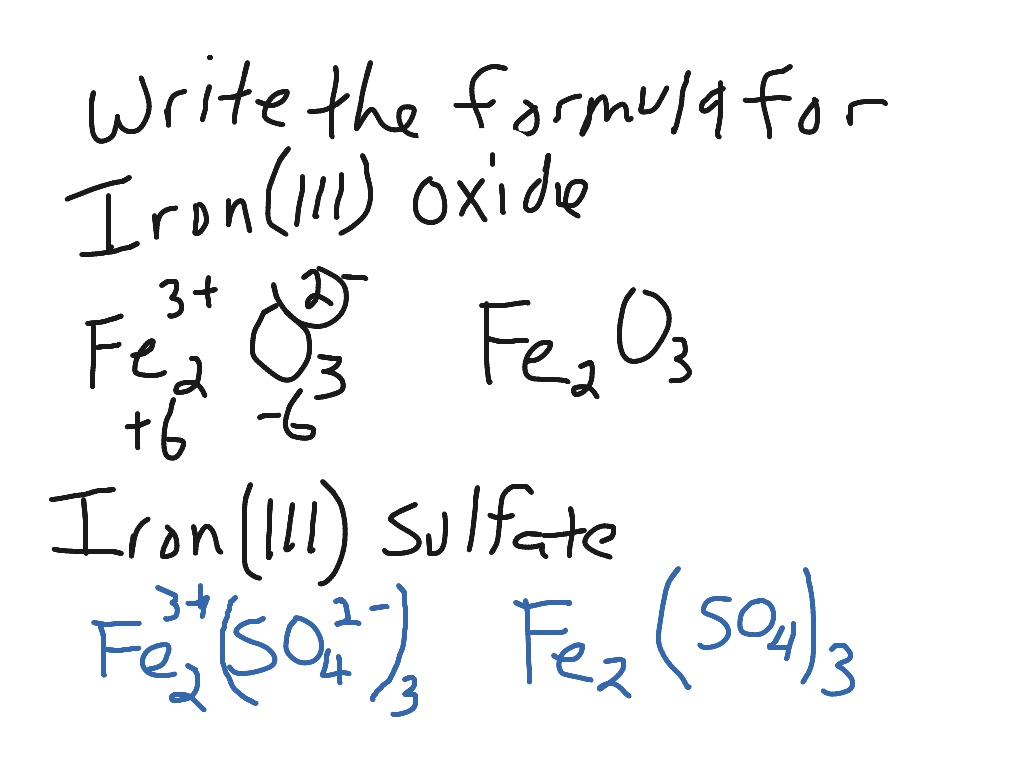SOLVED: "This the chemical formula for iron(II) hydroxide: Fe(OH) 2' Calculate the mass percent of oxygen in iron(II) hydroxide- Round your answer to tne nearest percentage-"

Negative ion Formula in compoundcharge Oxide Hydroxide Nitrate NO 3 -1 Sulphate Carbonate. - ppt download
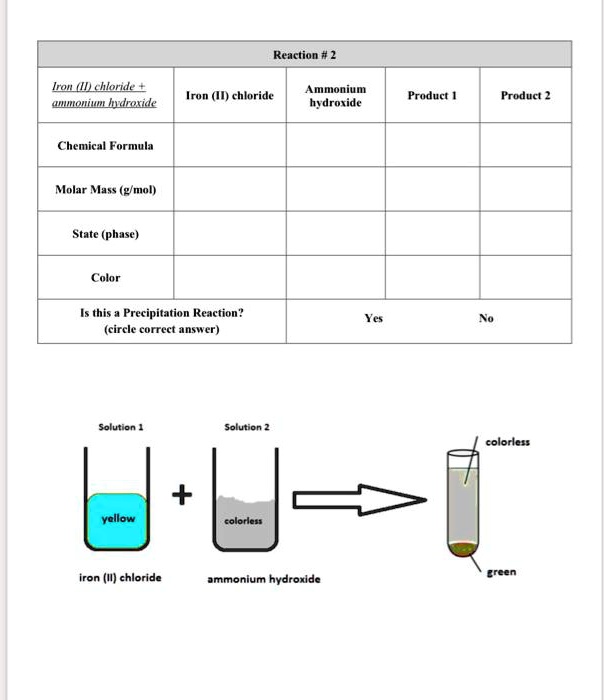
SOLVED: Reaction #2 Iron(II) chloride Ammonium hydroxide Iron(I) chloride Product Product 2 Chemical Formula Molar Mass (g/mol) State (phase) Color Is this a Precipitation Reaction? (circle correct answer) Yes Solution Solution Color